- Blog
- Omnisphere 2 crack error loading waveform
- Play free spider solitaire 2 suits
- Quest diagnostics fresno appointment
- Rr cars on farming simulator 19
- Periodic table valence electrons and charges
- Krunker io aimbot
- Generally accepted accounting principles gaap pronunciation
- Classic wooden runabout boats for sale
- Download game ufc 3 pc full version
- Return to castle wolfenstein setup download pc
- Lccc eagles eye
- Game action pc free download
- British family tree queen victoria
- Population density map of the us
- Pubg pc download windows 10
- 3d pinball space cadet 3d pinball game
- Sexual positions for pregnancy
- Blue yeti microphone drivers download windows 10
- Empire total war youtube darthmod install guide
- Flipnote studio 3d download code generator
- Ghost of tsushima mythic tales map
- Attack on titan game free online
- Initial d psp iso
- Respondus lockdown browser and covering my camera
- Cuphead free online games no download
- Detroit metro airport map southwest airlines
- Filemaker pro 12 trial download
- Original bard-s tale download for windows 10
- Internet connectivity test
- Tico tico no fuba sheet music piano
- List of free safe porn sites
- Motorcycle ecu flash price
- Sims 4 cc bts albums
- Susan ann reames in santa monica
- Macros meal planner app
- Windows 10 pro pirates bay
- Set timer 1 hour 14 minutes
- Just crack omnisphere 2 windows
- Adobe after effects cc 2014 serial number
- M audio usb midisport 2x2 driver
- The chaperone comic 3d
- Playspace mover index
- Boy scout meal planning sheet
- Cursive tattoo font alphabet
For the transition metals with partially-filed d shells, valence electrons are those electrons outside the noble gas core. Valence electrons are outer shell electrons for main group elements. This is because as Z increases by a small interval, the shell number increases, and so the electrons in the valence shell are much farther from the nucleus and are more shielded by all the electrons in the lower shell numbers. In chemistry and physics, a valence electron is an electron associated with an atom that can form a chemical bond and participate in a chemical reactions. This phenomenon is due to the opposite charges on each ion.Ĭovalent bonding is the sharing of electrons between atoms.\( \newcommand\) (from Ne to Na, for example). Most ionic compounds tend to dissociate in polar solvents because they are often polar. At the ideal interatomic distance, attraction between these particles releases enough energy to facilitate the reaction. It goes much deeper into the concept and delivers an extensive view of concepts like valence, charges, element names, etc. This information is available on a Color Periodic Table of the Elements or a Black and White version.

However, this reaction is highly favorable because of the electrostatic attraction between the particles. Our periodic table printable PDF does not contain the names of the elements only. It is an icon of chemistry and is widely used in physics and other sciences. The predicted overall energy of the ionic bonding process, which includes the ionization energy of the metal and electron affinity of the nonmetal, is usually positive, indicating that the reaction is endothermic and unfavorable. The periodic table, also known as the periodic table of the elements, is an ordered arrangement of the chemical elements into rows ('periods') and columns ('groups'). This chlorine atom receives one electron to achieve its octet configuration, which creates a negatively charged anion. This creates a positively charged cation due to the loss of electron. This sodium molecule donates the lone electron in its valence orbital in order to achieve octet configuration. In ionic bonds, the net charge of the compound must be zero.

Noble gases have stable valence electrons, halogens are very reactive with 7 valence electrons, nonmetals are extremely reactive, while metals conduct heat and. The charges on the anion and cation correspond to the number of electrons donated or received. The periodic table contains elements with different properties, such as metals and alkaline earth metals, each with specific valence electrons and charges. In ionic bonding, more than 1 electron can be donated or received to satisfy the octet rule. Similarly, nonmetals that have close to 8 electrons in their valence shells tend to readily accept electrons to achieve noble gas configuration. Comprehensive Information: The printable periodic table with names, charges, and valence electrons offers a comprehensive overview of each element. By losing those electrons, these metals can achieve noble gas configuration and satisfy the octet rule. According to Coulombs law, the attraction of an electron to a nucleus depends only on three factors: the charge of the nucleus (+Z), the charge of the electron (-1), and the distance between the two ((r)). Ionic bonding is observed because metals have few electrons in their outer-most orbitals. The ideal gas law is easy to remember and apply in solving problems, as long as you get the proper values a. Ionic bonds require an electron donor, often a metal, and an electron acceptor, a nonmetal.
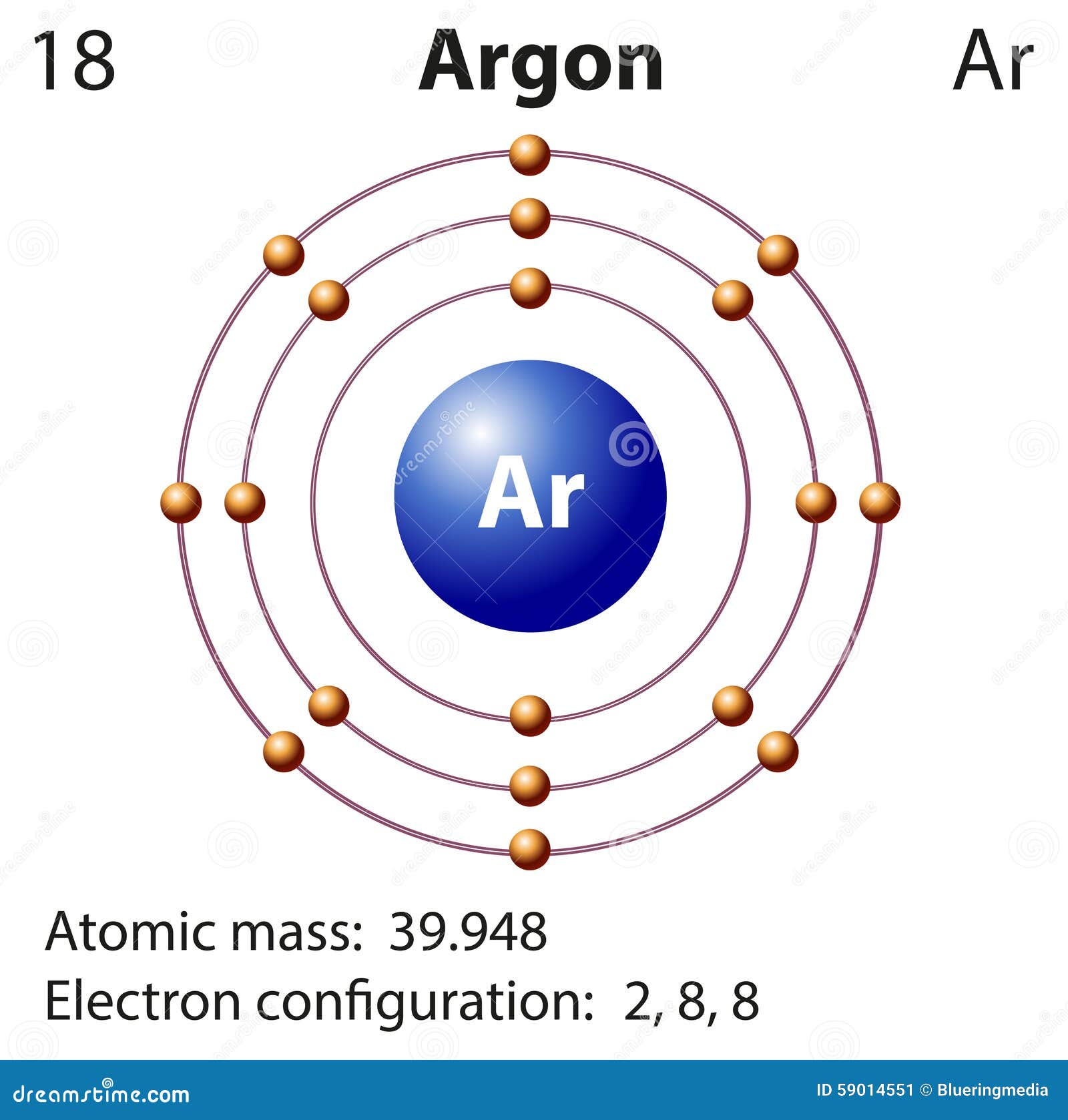

Atoms are electrically neutral because the number of protons, which carry a 1+ charge, in the nucleus of an atom is equal to the number of electrons, which carry a 1- charge, in the atom. Image credit: Wikipedia Commons, public domain. The attraction of oppositely charged ions is what makes ionic bonds. Sodium chloride is an ionic compound made up of sodium ions and chloride ions in a crystal lattice. This periodic table in PDF format can be downloaded for printing. This table also contains the element number, element symbol, element name, and atomic weights of each element. Atoms have a tendency to have eight electrons in their valence shell. This printable periodic table contains the atomic number, element symbol, element name, atomic weights and most common valence charges. Valence electrons determine the reactivity of an atom. In ionic bonds, the metal loses electrons to become a positively charged cation, whereas the nonmetal accepts those electrons to become a negatively charged anion. Valence electrons are the outer-shell electrons of an atom. It is a type of chemical bond that generates two oppositely charged ions. Ionic bonding is the complete transfer of valence electron(s) between atoms.

